Neda Daukšaitė1, Gintarė Valterytė1, Laura Marcinkienė3, Vytautas Zabiela2, Rita Kupčinskaitė-Noreikienė3, Diana Žaliaduonytė2
1Medical Academy, Lithuanian University of Health Sciences, Kaunas, Lithuania.
2Department of Cardiology, Medical Academy, Lithuanian University of Health Sciences,
Kaunas, Lithuania.
3Department of Hematology and Oncology, Medical Academy, Lithuanian University of Health Sciences, Kaunas, Lithuania.
Abstract
1.1 Background
Primary tumors of the pericardium are rare. Secondary or metastatic pericardial disease is much more common. Symptomatic pericarditis may be the first clinical manifestation of malignancy. The diagnosis is usually an incidental finding during imaging processing. Pericardiocentesis and percutaneous drainage of pericardial effusion (PE) are indicated in PE if neoplastic etiology is suspected. In this report, we describe the case of a patient presenting with nonspecific symptoms of PE and neoplastic disease.
1.2 Case presentation
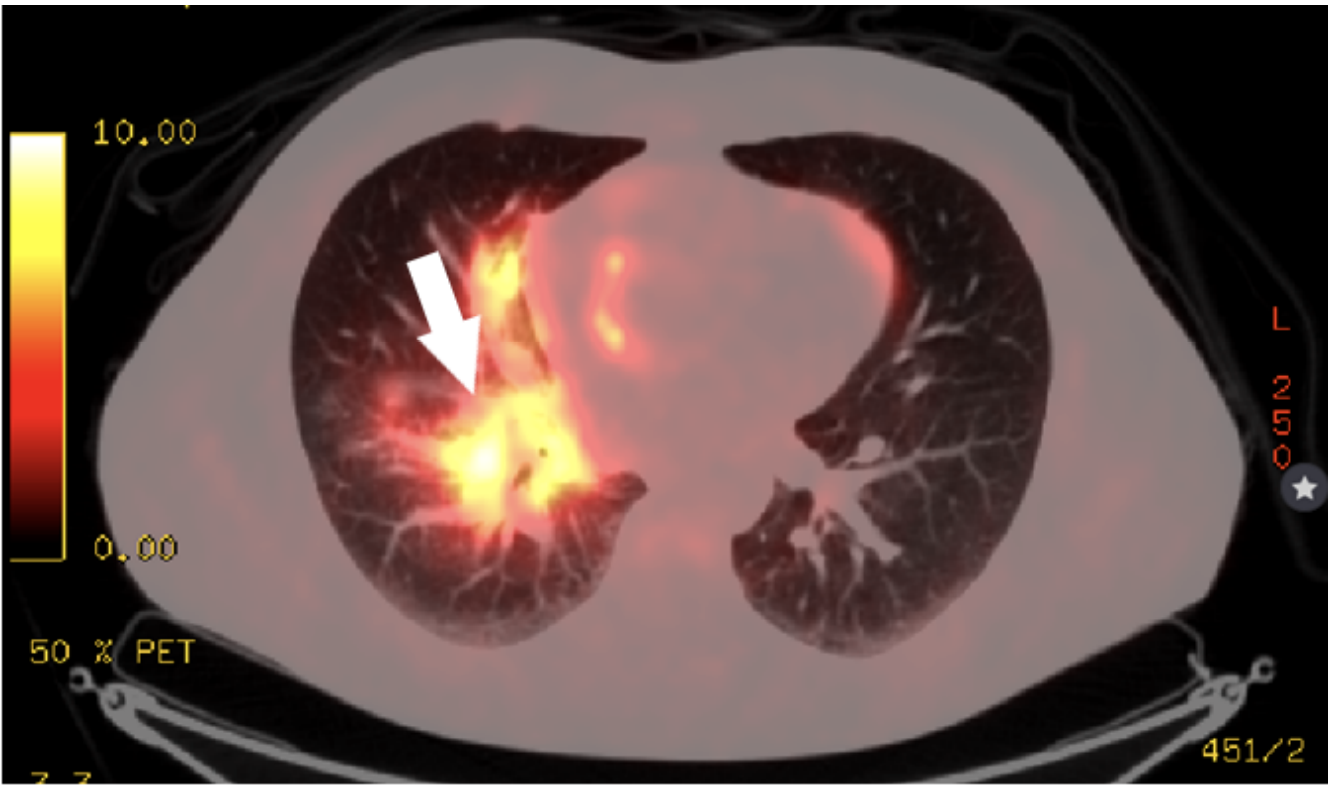
A 47 – year old man was admitted to the hospital with 2 years history of intermittent low – grade fever, non – productive cough, malaise, and dyspnea. Computed tomography (CT) scan was done allegedly to find the pulmonary embolism, but the CT scan disclosed multiple enlarged lymph nodes in mediastinal, intraperitoneal areas, and pericardial effusion. Pericardial effusion and neoplastic disease were diagnosed by lymph node biopsy and positron emission tomography/computed tomography (PET/CT), respectively. Treatment started with Alectinib and radiation therapy. However, after 10 months the progression of the disease was observed, so treatment changed to second-line drug Lorlatinib. In order to prevent pathological bone fractures, treatment changed to Denosumab. It resulted in the complete remission of PE. In 1 year and 6 months after diagnosis of the lung cancer was confirmed regarding the successful treatment, the patient is fully active, back to social life with no signs of dyspnea.
1.3 Conclusions
The diversity of clinical manifestation (such as low – grade fever, non – productive cough, malaise, dyspnea, and PE) in such a potentially severe disease should alert the physician to prompt diagnosis and treatment of malignant process.
1.4 Keywords: Pericarditis; Effusion; Neoplastic disease; Case; Malignancy.